Supor® grade EAV membrane filters are designed for effective bioburden and particle control of buffers and pharmaceutical or biological process fluids. A highly asymmetric single layer membrane incorporated in these filters ensures high throughputs and flow rates when utilized for the protection of downstream chromatography columns, ultrafilters or sterilizing filters.
Supor grade EAV membrane filters allow for reduced sizing of filter systems with improved process efficiencies when use of validated sterilizing grade filters is not essential, but reliable bioburden control is required. Supor grade EAV membrane filters are also effective prefilters for protection and extending the life of 0.2 µm sterilizing-grade and finer membrane filters where required.
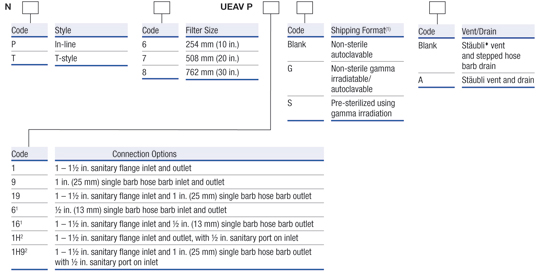
Pall’s range of Kleenpak™ Nova capsules are designed for use in medium to large scale production environments (100 L > 1000 L), often selected by the end user following scaling studies using smaller Kleenpak capsule formats. With the AB style cartridge format at its core, this capsule filter style can be supplied with the most comprehensive range of filter media.
Features and Benefits
- Hydrophilic, controlled asymmetric polyethersulfone (PES) membrane ensures high microbial and particulate reduction with outstanding service life
- Bacterial titer reduction in excess of 6 log for Brevundimonas diminuta ensures low bioburden levels in filtrate, regardless of bacteria or particle loading
- Broad pH compatibility for processing a wide range of buffers and other fluids
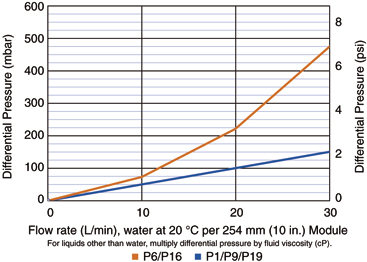
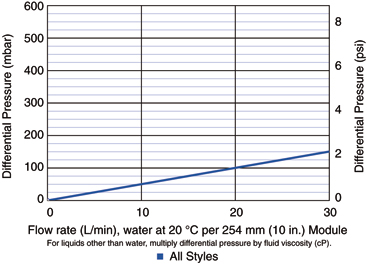
- High membrane area featuring Pall-patented cresent-shaped laid-over pleat construction combined with a narrow diameter core guarantees high flow rates, robustness and smaller multi-cartridge assemblies
- Kleenpak™ capsule formats eliminate housing cleaning and associated validation, for ease of use and integration into Allegro single-use disposable systems
- Low-binding polyethersulfone membrane for maximum transmission of proteins
Quality Standards
- 100% integrity tested
- Manufactured for use in conformance with cGMP
- Each filter is fully traceable by individual marked lot and serial number
- Manufactured under an ISO 9001 Certified Quality System
- Meets USP Biological Reactivity Test, in vivo, for Class VI-121 °C Plastics
- Certificate of Test provided confirms:
- Fabrication Integrity
- Bacterial Retention
- Materials of construction
- Effluent quality for cleanliness, TOC, water conductivity, pH and pyrogens